This site is intended for United Kingdom/Ireland healthcare professionals
Decision-making factors in AML:
Insights from BSH 2022
Treatment selection and care for patients with AML can be challenging, so we spoke to three experts in the field about their practice and the factors they consider when making choices for, and with, their patients.

Explore the questions and watch the videos below to find out what drives decision-making for Dr Victoria Campbell, Dr Steven Knapper and Dr Priyanka Mehta
The role of real-world
evidence in clinical
practice
Answer two quick questions and watch the video
The role of real-world evidence in clinical practice
Question 1
What proportion of patients received an HSCT following treatment with Vyxeos Liposomal in the French real-world study?
Please select an answer
 Incorrect - 35%
Incorrect - 35%Patients received an HSCT in each study at the following rates:
France RWE: 35%1
England RWE: 25%2
Germany RWE: 62%3
Italy RWE: 40% of those who achieved CR/CRi4
Patients received an HSCT in each study at the following rates:
France RWE: 35%1
England RWE: 25%2
Germany RWE: 62%3
Italy RWE: 40% of those who achieved CR/CRi4
Question 2
Real-world studies complemented the mOS data for Vyxeos Liposomal from Study 301. What range do these results sit within?
Please select an answer
 Incorrect - 16 months - NR
Incorrect - 16 months - NRFrance RWE: 16.1 months1
England RWE: 16.6 months2
Germany RWE: 21 months3
Italian RWE: mOS NR - 68.6% at 12 months4
France RWE: 16.1 months1
England RWE: 16.6 months2
Germany RWE: 21 months3
Italian RWE: mOS NR - 68.6% at 12 months4
The role of real-world evidence in clinical practice
Watch as AML experts Steven Knapper and Victoria Campbell discuss their key takeaways from the available studies.
Selecting a treatment
approach: Intensive vs
non-intensive chemotherapy
Answer two quick questions and watch the video
Selecting a treatment approach: Intensive vs non-intensive chemotherapy
Question 1
Based on the ASH 2020 guidelines, which is the suggested treatment for older adults with AML who are not eligible for HSCT?
Please select an answer
 Incorrect - Intensive anti-leukaemic therapy followed by post-remission therapy
Incorrect - Intensive anti-leukaemic therapy followed by post-remission therapyRecommendation 1
Anti-leukaemic therapy over best supportive care5
Recommendation 2
Intensive anti-leukaemic therapy over less-intensive anti-leukaemic therapy5
Recommendation 3
Patients who achieve remission following at least one cycle of intensive anti-leukaemic therapy should receive allogeneic HSCT if elligible. ASH guidelines suggest ineligible patients should receive postremission therapy over no additional therapy5
Recommendation 1
Anti-leukaemic therapy over best supportive care5
Recommendation 2
Intensive anti-leukaemic therapy over less-intensive anti-leukaemic therapy5
Recommendation 3
Patients who achieve remission following at least one cycle of intensive anti-leukaemic therapy should receive allogeneic HSCT if elligible. ASH guidelines suggest ineligible patients should receive postremission therapy over no additional therapy5
Question 2
Which clinical factors should be considered when selecting a treatment option for patients with AML-MRC/t-AML?
Please select an answer
 Incorrect - All of the above
Incorrect - All of the aboveECOG PS, cytogenetic risk, comorbidities, and age are all among the factors to be considered in patient assessment and treatment selection7-9
ECOG PS, cytogenetic risk, comorbidities, and age are all among the factors to be considered in patient assessment and treatment selection7-9
Selecting your treatment approach: Intensive vs non-intensive chemotherapy
Experts share the factors that affect their choice to administer intensive or non-intensive chemotherapy to AML-MRC and t-AML patients.
The value of supportive
care in IC: Managing
myelosuppression
Answer two quick questions and watch the video
The value of supportive care in IC: Managing myelosuppression
Question 1
Based on the data from Study 301, how much longer does neutrophil and platelet recovery take for Vyxeos Liposomal patients vs those on DA 3+7?
Please select an answer
 Incorrect - 6-8 days
Incorrect - 6-8 daysMedian time to platelet and neutrophil recovery is 6-8 days longer with Vyxeos Liposomal vs DA 3+76
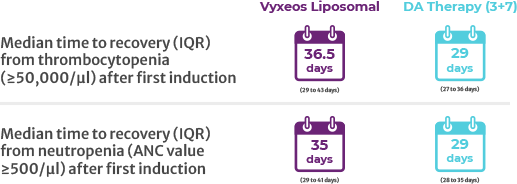
Median time to platelet and neutrophil recovery is 6-8 days longer with Vyxeos Liposomal vs DA 3+76

Question 2
True or false: Prolonged myelosuppression with Vyxeos Liposomal vs DA 3+7 also translated into higher early mortality rates?
Please select an answer
 Incorrect - False
Incorrect - FalseDay 30 and Day 60 mortality rates were lower with Vyxeos Liposomal vs DA 3+7:
France RWE: 30-day – 6%, 60-day – 8%1
England RWE: 30-day – 7%, 60-day – 15%2
Germany RWE: 30-day – 8%3
Italian RWE: 60-day – 7%4
Study 301: Vyxeos Liposomal 30-day - 6%, 60-day - 14% vs DA 3+7 30-day - 11%, 60-day 21%6
Day 30 and Day 60 mortality rates were lower with Vyxeos Liposomal vs DA 3+7:
France RWE: 30-day – 6%, 60-day – 8%1
England RWE: 30-day – 7%, 60-day – 15%2
Germany RWE: 30-day – 8%3
Italian RWE: 60-day – 7%4
Study 301: Vyxeos Liposomal 30-day - 6%, 60-day - 14% vs DA 3+7 30-day - 11%, 60-day 21%6
The value of supportive care in IC: Managing myelosuppression
Experts share their experience of treating patients with Vyxeos Liposomal as well as advice on how myelosuppression can be managed following IC.
Championing patient care
by making the most of the
AML community
Answer two quick questions and watch the video
Championing patient care by making the most of the AML community
Question 1
What type of support would you find most useful?
Please select an answer
Question 2
What type of learning events do you prefer?
Please select an answer
Championing patient care by making the most of the AML community
Experts share their tips and advice on forging a successful AML treatment practice.
Abbreviations
AML, acute myeloid leukaemia; AML-MRC, acute myeloid leukaemia with myelodysplasia-related changes; ASH, American Society of Hematology; BSH, British Society for Haematology; CR, complete response; CRi, complete response with incomplete platelet or neutrophil recovery; DA, daunorubicin + cytarabine; ECOG, Eastern Cooperative Oncology Group; HMA, hypomethylating agent; HSCT, haematopoietic stem cell transplantation; IC, intensive chemotherapy; IQR, interquartile range; LDAC, low-dose cytarabine; mOS, median overall survival; NR, not reached; PS, performance status; RWE, real-world evidence; t-AML, therapy-related AML
References
- Chiche E, et al. Blood Adv 2021;5(1):176–184
- Legg A, et al. Blood 2021; 138 (Suppl.1): 2310
- Rautenberg C, et al. Blood Cancer J 2021;11:164
- Guolo F, et al. Blood Cancer J 2020;10(10):96
- Sekeres M, et al. Blood Adv 2020;4(15):3528–3549
- Lancet JE, et al. J Clin Oncol 2018;36(26):2684–2692
- Wheatley K, et al. Br J Haematol 2009;145(5):598–605
- Deschler B, et al. Haematologica 2013;98(2):208–216
- Ferrara F, et al. Leukemia 2013;27(5):997–999
